 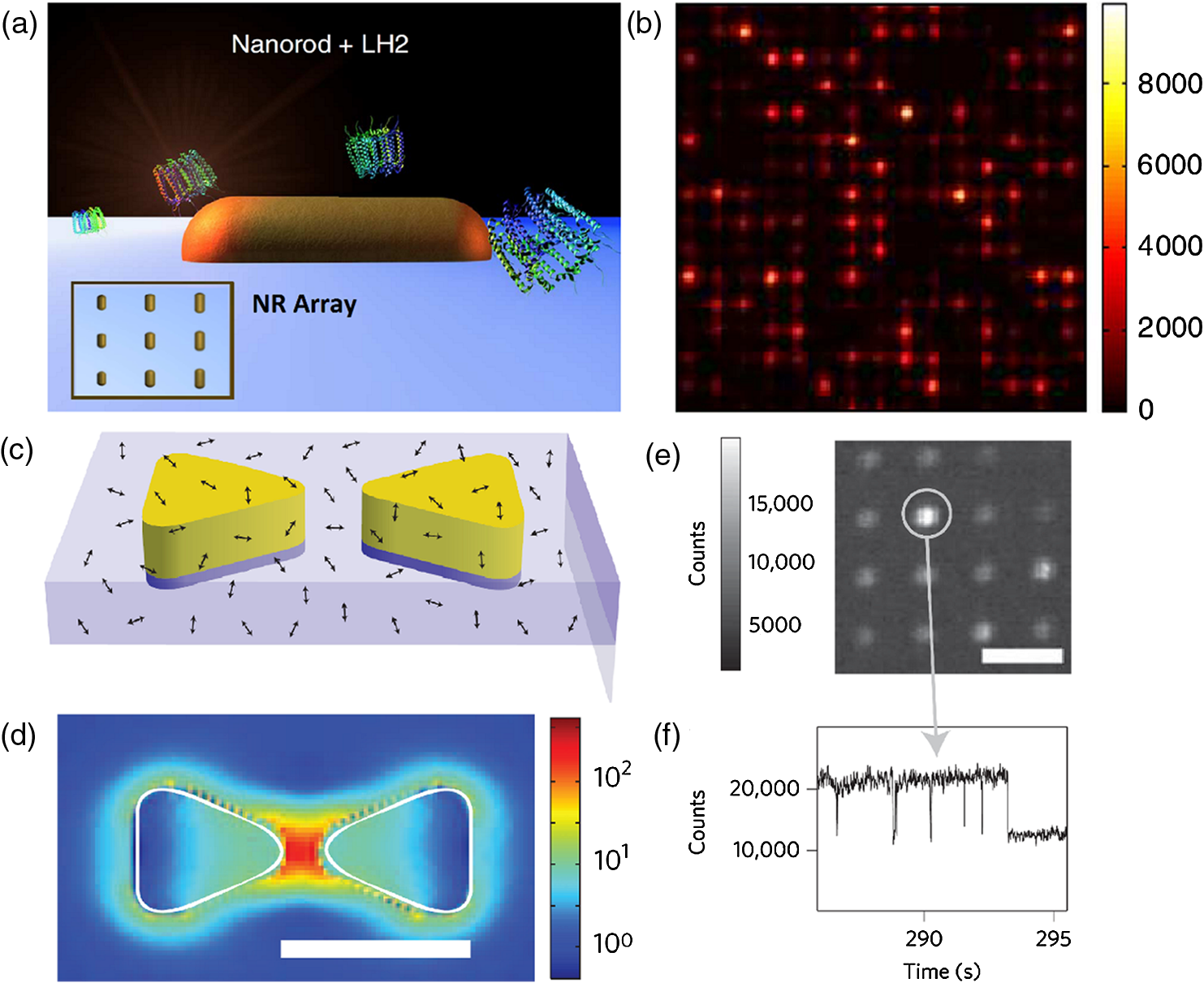
Moving from row to row may result in molecules remaining excited from the previous zap, resulting in a noisy image. These pulses can either be activated in rows or can be randomized, normally being activated for only 100 picoseconds or less. Pulsing these lasers in multiple random regions will create a high-resolution image of the sample that is being examined. 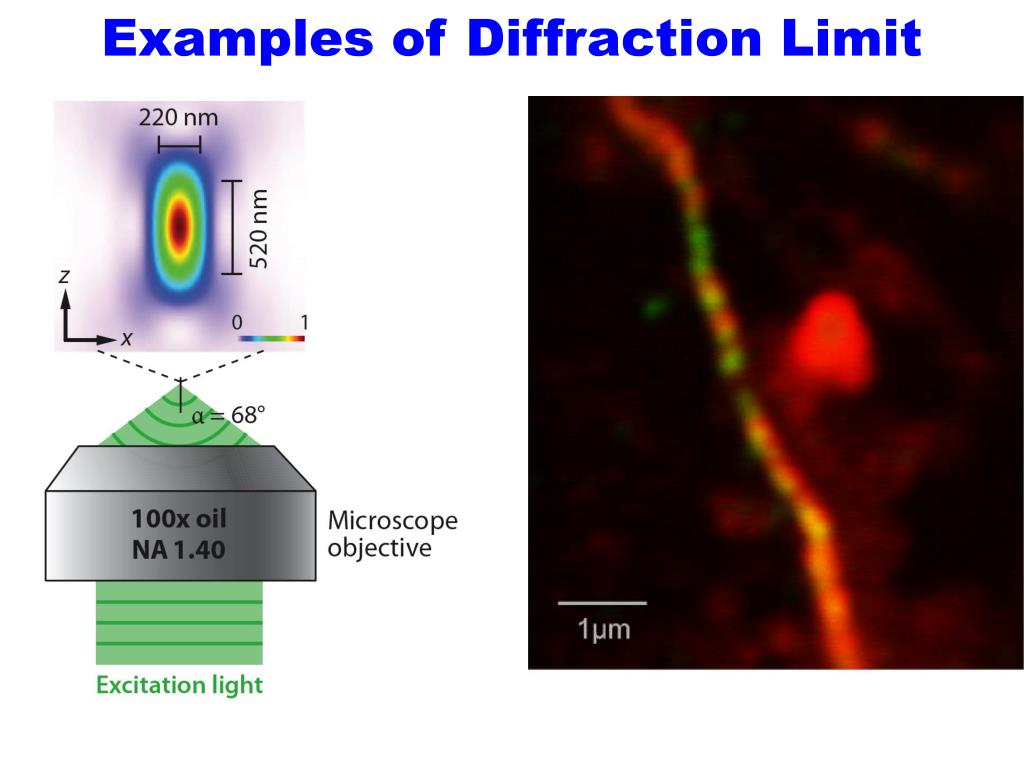
Pulsed STED is where the excitation and redshifted light are emitted right after one another, this is how STED was first introduced. Scanning a large area with successive beams will result in a large image once all the sections are captured and stitched together. The donut shape of the second beam can be adjusted in size to accommodate the size of the sample that is being absorbed. These lasers are switched in picosecond time frames, capturing areas with extreme precision. This allows the microscope to see the finest of details. This second beam will turn “off” the molecules in the region, allowing the detector to capture only the region of the first excitation, where the molecule will be glowing due to the fluorescent light. A secondary laser, called a depletion laser, is then activated shortly after the first beam this wavelength is red-shifted compared to the primary laser and will create a donut around the first beam. One wavelength at the level where the sample will absorb the energy and reemit it as fluorescent light. A laser is pointed at certain areas of interest where it will emit two different wavelengths. STED microscopy utilizes the absorption and emission of fluorescent dyes. Image sourced from the article: High-Resolution 3D Light Microscopy with STED and RESOLFT. How confocal microscopy works and the diffraction barrier equation associated with it. This is achieved by minimizing the focal point illuminated by the excitation light, and thus enhancing the achievable resolution. STED microscopy allows bypassing the diffraction limit by creating super-resolution images by the selective deactivation of fluorophores. This relationship is also known as the Abbe’s diffraction limit. n*Sin(a) is the numerical aperture (N.A.) of the objective/lens. As shown on Figure 1, the Abe equation for the minimum resolvable distance between features is given by the average wavelength over the quantity of two times n (refractive index) and sin(acceptance angle or angle of view). Modern equipment can reach a numerical aperture of roughly 1.4 using immersion technology (where the medium between the objective lens and the sample is immersed in fluid medium with refractive index higher than that of air), leading to a lateral resolution of around 250nm. Using shorter wavelength of light and a larger numerical aperture of the lens, a diffraction limited performance can be achieved for visible microscopes. The performance limit of such microscopes is determined by the diffraction limited performance of the lenses used in the microscope. Visible light microscopy uses, just as the title suggests, visible light in the 400-700nm range. Visible Light Microscopy Coming To An End? Visible Light Microscopy Coming To An End?.In this article, we will briefly go over these topics of STED microscopy. Using both an excitation and depletion laser, researchers can target dimensions smaller than 200 nm within a cell and create a high-resolution image with multiple sample images. STED has solid reliability in the biological field where scientists have to observe living cells that emit fluorescence light. This is because STED microscopy leaps over the issues revolving around the diffraction limit that cripple regular microscopes. This method is superior to conventional light microscopy as the images have less noise and are sharper given that the inspected molecule is a living organism. Stimulated emission depletion microscopy (STED) is a popular super-resolution microscopy technique that researchers use to magnify microscopic organic compounds and proteins. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
